Protein intake after concurrent exercise: soy, whey and the role of leucine
What role does the protein source, and in particular the amino acid leucine, play in muscle building?

Title of publication: Myofibrillar and Mitochondrial Protein Synthesis Rates Do Not Differ in Young Men Following the Ingestion of Carbohydrate with Whey, Soy, or Leucine-Enriched Soy Protein after Concurrent Resistance- and Endurance-Type Exercise
Authors: Churchward-Venne, T. A., Pinckaers, P. J. M., Smeets, J. S. J., Peeters, W. M., Zorenc, A. H., Schierbeek, H., Rollo, I., Verdijk, L. B., & van Loon, L. J. C.
Journal: The Journal of nutrition, 149(2), 210–220.
Year: 2019
In the previous blog post, we showed how milk protein accelerated the rate at which new protein structures are formed in muscle after concurrent training. Within the field of muscle protein metabolism, an interesting concept exists: the leucine threshold (1). A protein consists of smaller building blocks called amino acids. There are a total of 20 different amino acids found in a protein structure. One of these is the amino acid leucine. And leucine is a special amino acid because, independently of the other 19 amino acids, it can trigger the muscle protein synthesis pathway via the activation of the transcription factor mammalian target of rapamycin (mTOR). But! One leucine molecule doesn't suddenly activate the pathway. Neither does two leucine molecules. No, a certain amount and a certain rate of digestion are required before the floodgates actually open: the leucine threshold.
Activation pathway what?
In biology, cells operate based on signals and stimuli, and messages are constantly being transmitted within the cell and between cells. An external stimulus such as exercise can cause a cell to respond to this disruption of the status quo. This stimulus then activates many messages. The response to such a message is often the activation of a cellular process. For example, a muscle adapts to exercise:
- Stimulus that causes disruption (exercise)
- Molecular pathways (message deliverers) are activated to deliver a signal to the cell's main regulator: DNA in the nucleus.
- The message is read and translated into an action.
- The cell begins to build new structures to adapt to the disruption. In this case, muscle protein synthesis, so that in the future, the cell has adapted to better cope with the same disruption: becoming stronger.
Below you see a complex diagram showing how activation of target of rapamycin (mTOR) by muscle contraction and leucine leads to a boost in the process of muscle protein synthesis.
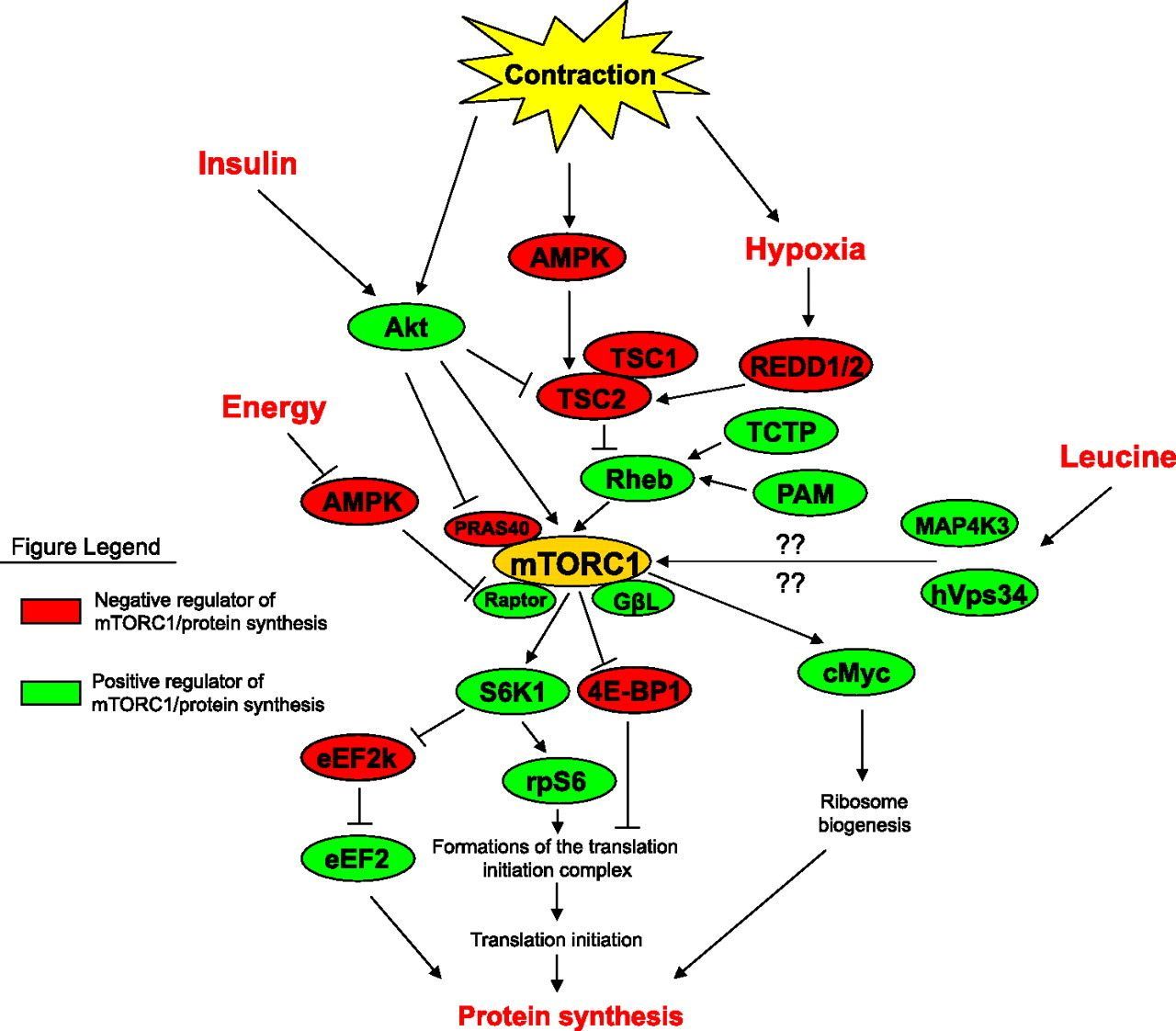
Illustration by: Drummond, M. J., Dreyer, H. C., Fry, C. S., Glynn, E. L., & Rasmussen, B. B. (2009). Nutritional and contractile regulation of human skeletal muscle protein synthesis and mTORC1 signaling. Journal of applied physiology (Bethesda, Md. : 1985), 106(4), 1374–1384. https://doi.org/10.1152/japplphysiol.91397.2008
Background:
All proteins have different compositions and therefore different amounts of leucine. Animal proteins generally contain higher concentrations of leucine than plant proteins (2). How this translates to the activation of muscle protein synthesis has been demonstrated in several studies. For example, consuming plant proteins (soy) resulted in a lower stimulation of MPS compared to whey (3). Another study showed that consuming a small amount of whey (6 grams) but enriching (adding) it with the amino acid leucine to the concentration found in much higher doses of whey (25 grams) induces a similar increase in muscle protein synthesis after resistance training as consuming just 25 grams of whey (4). Because soy is an "inferior" dietary protein for stimulating muscle protein synthesis (MPS) due to its lower leucine concentration, the theory is that adding leucine to soy to match the amount in whey would result in a similar increase in MPS. So that's what we tested in this study, but again after a competitive training session instead of just strength training.
What we did:
The same protocol was used as in the previous blog post, but only the recovery drinks the subjects received were different: again, everyone consumed 45 grams of carbohydrates after training. In addition, one group consumed whey protein (20 grams) (the group from the previous study). Another group consumed soy protein (20 grams), and the last group consumed 20 grams of soy, but we added an amount of free leucine to this drink that matched the amount of leucine in 20 grams of whey.
What we found:
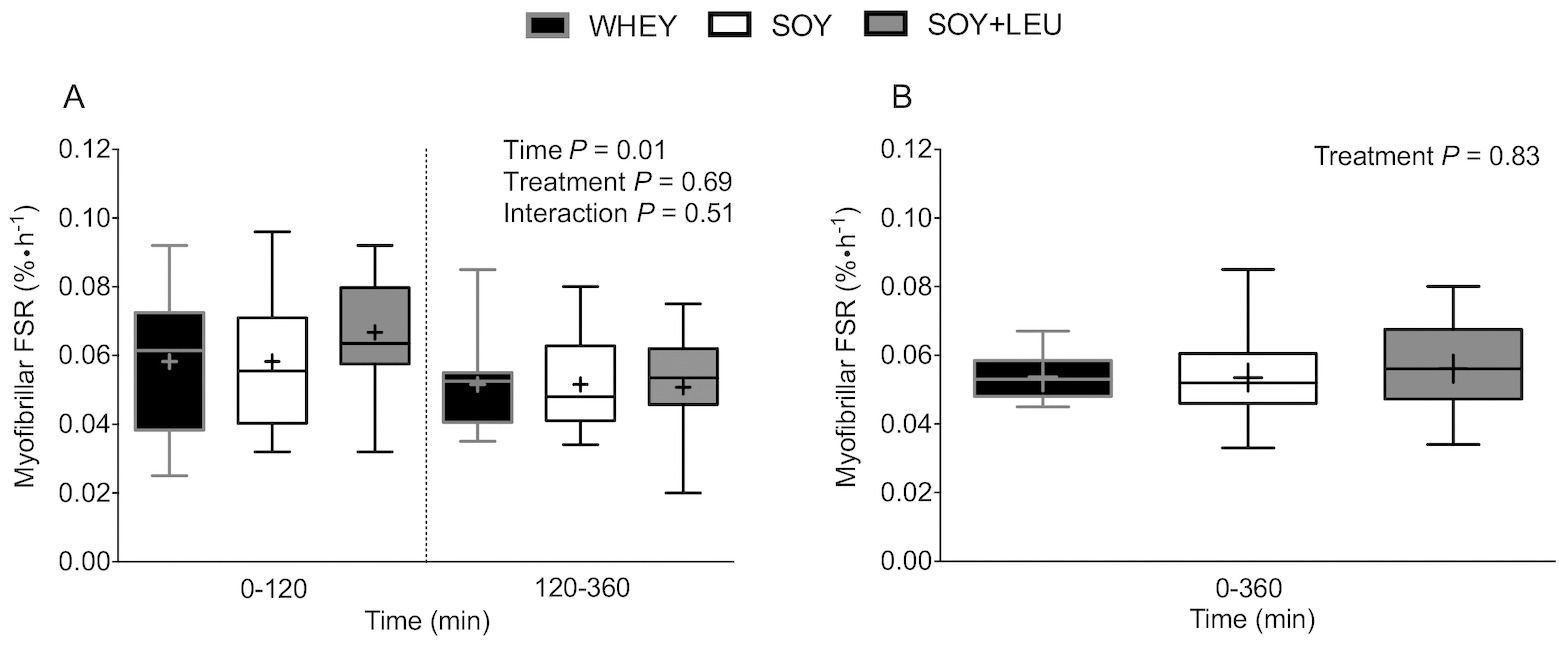
Here too, we measured the rate of both myofibrillar and mitochondrial protein synthesis, as well as mTOR activation. When comparing the groups, no differences were found in myofibrillar and mitochondrial protein synthesis.
What does this mean?
These findings were somewhat surprising based on previous observations, and there was no clear explanation for the observation that soy without leucine enrichment yielded similar MPS values as whey and soy with leucine enrichment. It was "reassuring" that the activation pathway was also similar, but the amount of leucine in the blood differed between the high-leucine and low-leucine groups. Perhaps the protein dose or the difference in leucine levels was not high or large enough to observe an effect? Perhaps the type of training (competitive training compared to the previous experiments with only resistance training) influenced the outcome? Perhaps the leucine threshold hypothesis is not as rigid as initially thought? Science is actually quite fun, especially when the results don't support your previous hypothesis and the outcome of an experiment raises more questions than answers!
Personal reflections:
This publication arose from the same experiment as in the previous blog post. To expand on my previous personal reflections: this master's project taught me to do something that sparks enthusiasm and passion. When I was deciding which master's project to choose, I was also in discussions with a professional cycling team, which was very important to me, as that's where I ultimately wanted to work. However, the team was realistic about what they could offer, which mainly consisted of going through stacks of Excel spreadsheets to analyze data. This didn't generate any enthusiasm at the time, so the other project was chosen. However, it did open up a whole new world and ignited my passion for sports nutrition, so I think I made the right decision and that I...
Reference list:
(1) Zaromskyte, G., Prokopidis, K., Ioannidis, T., Tipton, K. D., & Witard, O. C. (2021). Evaluating the Leucine Trigger Hypothesis to Explain the Post-prandial Regulation of Muscle Protein Synthesis in Young and Older Adults: A Systematic Review. Frontiers in nutrition, 8, 685165. https://doi.org/10.3389/fnut.2021.685165
(2) Gorissen, S. H. M., Crombag, J. J. R., Senden, J. M. G., Waterval, W. A. H., Bierau, J., Verdijk, L. B., & van Loon, L. J. C. (2018). Protein content and amino acid composition of commercially available plant-based protein isolates. Amino acids, 50(12), 1685–1695. https://doi.org/10.1007/s00726-018-2640-5
(3) Tang, J. E., Moore, D. R., Kujbida, G. W., Tarnopolsky, M. A., & Phillips, S. M. (2009). Ingestion of whey hydrolyzate, casein, or soy protein isolate: effects on mixed muscle protein synthesis at rest and following resistance exercise in young men. Journal of applied physiology (Bethesda, Md.: 1985), 107(3), 987–992. https://doi.org/10.1152/japplphysiol.00076.2009
(4) Churchward-Venne, T. A., Burd, N. A., Mitchell, C. J., West, D. W., Philp, A., Marcotte, G. R., Baker, S. K., Baar, K., & Phillips, S. M. (2012). Supplementation of a suboptimal protein dose with leucine or essential amino acids: effects on myofibrillar protein synthesis at rest and following resistance exercise in men. The Journal of physiology, 590(11), 2751–2765. https://doi.org/10.1113/jphysiol.2012.228833
Original publication link:







